Tim Cook answered
The chemical reaction between "C5H10 + HBr" is actually a little bit ambiguous...
HBr for example, can represent either the compound hydrogen bromide, or hydrobromic acid, formed when hydrogen bromide is dissolved in water.
A Possible Answer
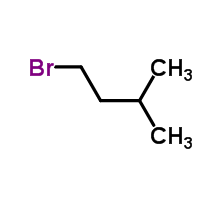
Having looked around, the outcome you are most likely looking for is C5H11Br, also known as Isoamyl Bromide or 1-Bromo-3-methylbutane (pictured). This highly flammable substance has no one particular use other than in the production of other compounds and pharmaceuticals, but was once used in spectrometry and as an extraction solvent.
If you're not to sure about the range of hydrocarbon isomers, this YouTube film might come in useful: