The chemical formula for acetic acid (pictured) is C2H4O2. The chemical formula for 1-Pentanol is C5H12O.
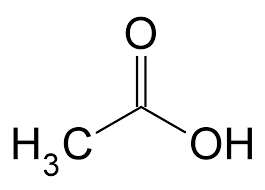
As their formulas suggest, an acetic acid molecule contains 2 carbon atoms, 4 hydrogen atoms and 2 oxygent atoms, while a molecule of 1-Pentanol comprises 5 carbon atoms, 12 hydrogen and 1 oxygen atom.
1-Pentanol, also known as amyl alcohol or pentyl alcohol, is used in the production of compact discs. It has a low burning point (49C) and can be hazardous to health if inhaled or spilled onto exposed skin, and so should be treated with caution.
Acetic acid, sometimes called ethylic acid, is a reagent and has more uses than 1-Pentanol, mainly in the production of vinyl acetate. Vinegar contains around 4% acetic acid, making it the second largest ingredient in vinegar apart from water, although its greatest use comes in the manufacture of other compounds.
Acetic acid is also one of the many organic compounds found in space!
1-Pentanol can also be used to make fruit flavorings and odors, as demonstrated in this film: